Loading history...
Atomic models for students

Summary of Atomic Models
| Model | Scientist | Year | Key Features | Limitations |
|---|---|---|---|---|
| Dalton | John Dalton | 1803 | Solid, indivisible spheres; atoms of same element identical | Couldn't explain electricity or how atoms combine |
| Thomson | J.J. Thomson | 1897 | Plum pudding model; electrons embedded in positive sphere | Didn't account for concentrated mass |
| Rutherford | Ernest Rutherford | 1911 | Nucleus with orbiting electrons; mostly empty space | Couldn't explain electron stability or energy |
| Bohr | Niels Bohr | 1913 | Electrons in fixed circular orbits at set energy levels | Only worked for hydrogen; didn't match observation |
| Schrödinger | Erwin Schrödinger | 1926 | Electron probability clouds (orbitals); wave-particle duality | Complex mathematics; abstract visualization |
The Atom: Building Block of Everything
Atoms are the fundamental building blocks of all matter in the universe. Everything you can see, touch, smell, or taste is made of atoms – from the air you breathe to the stars in the sky. Understanding atoms is the key to understanding chemistry, physics, and the nature of reality itself.
A Brief History of Atomic Discovery
Ancient Greek Philosophy (400 BCE)
Democritus and the "Atomos"
- Democritus: Greek philosopher who first proposed atomic theory
- "Atomos": Greek word meaning "uncuttable" or "indivisible"
- Basic idea: Matter cannot be divided forever
- Thought experiment: Cut a piece of matter repeatedly until it can't be cut anymore
Aristotle's Opposition
- Aristotle: Rejected atomic theory
- Four elements: Proposed earth, water, air, and fire instead
- Continuous matter: Believed matter was infinitely divisible
- Impact: Aristotle's views dominated for nearly 2000 years
John Dalton's Atomic Theory (1803)
The First Scientific Atomic Model
- Experimental basis: Based on chemical experiments and observations
- Key contributions: Made atomic theory scientific rather than philosophical
- Law of multiple proportions: Elements combine in whole number ratios
- Revolutionary idea: Different elements have different types of atoms
Dalton's Main Ideas
- All matter is made of tiny, indivisible particles called atoms
- Atoms of the same element are identical in mass and properties
- Atoms of different elements are different
- Atoms combine in simple whole number ratios to form compounds
- Atoms cannot be created, destroyed, or changed into other atoms
What made Dalton different from Democritus:
- Evidence-based: Dalton used experimental data, not just philosophy
- Quantitative: Assigned relative weights to different atoms
- Predictive: Could predict how elements would combine
- Testable: Other scientists could verify his conclusions
- Studied reactions of gases
- Measured combining ratios of elements
- Observed that compounds always had the same proportions
- Water: always 8 parts oxygen to 1 part hydrogen by mass
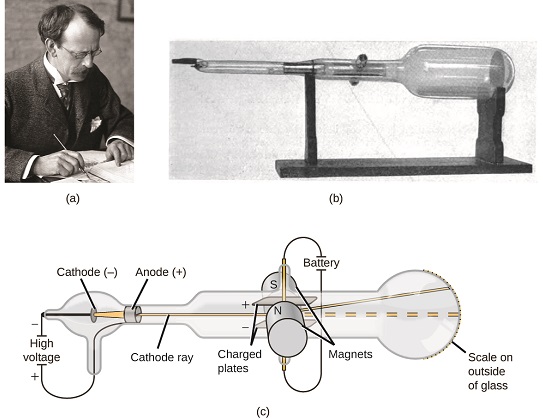
J.J. Thomson and the Electron (1897)
The Cathode Ray Experiments
- Cathode rays: Mysterious rays produced in vacuum tubes
- Thomson's discovery: These rays were streams of tiny particles
- Electron: First subatomic particle discovered
- Charge-to-mass ratio: Thomson measured this precisely
The Plum Pudding Model
- Structure: Atoms are spheres of positive charge
- Electrons: Embedded like plums in a pudding (or raisins in a cake)
- Distributed evenly: Negative electrons spread throughout
- Key insight: Atoms are NOT indivisible – they contain parts!
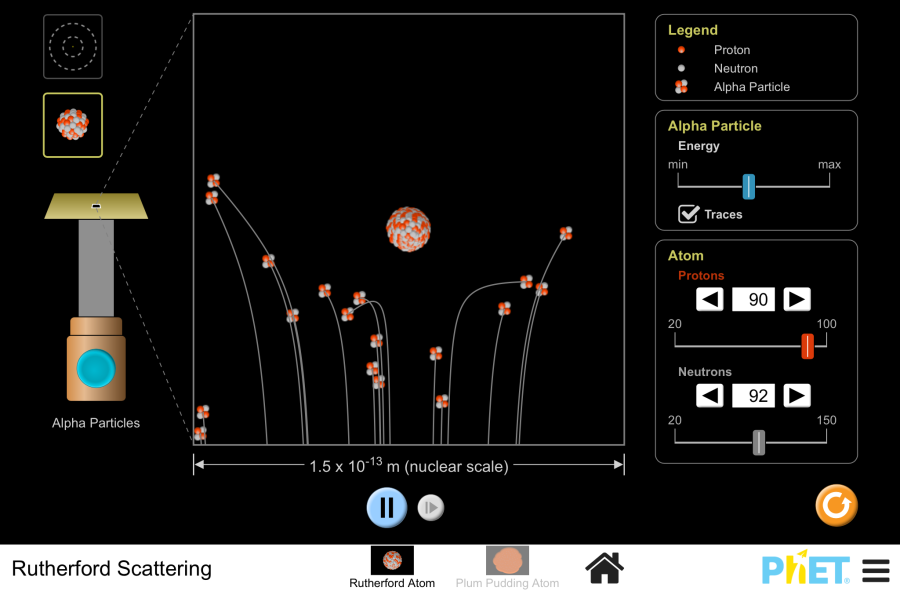
Ernest Rutherford and the Nucleus (1911)
The Gold Foil Experiment
- Setup: Alpha particles fired at thin gold foil
- Expected result: All particles pass straight through (if plum pudding model correct)
- Actual result: Most passed through, but some bounced back!
- Rutherford's reaction: "As if you fired a cannon at tissue paper and it bounced back"
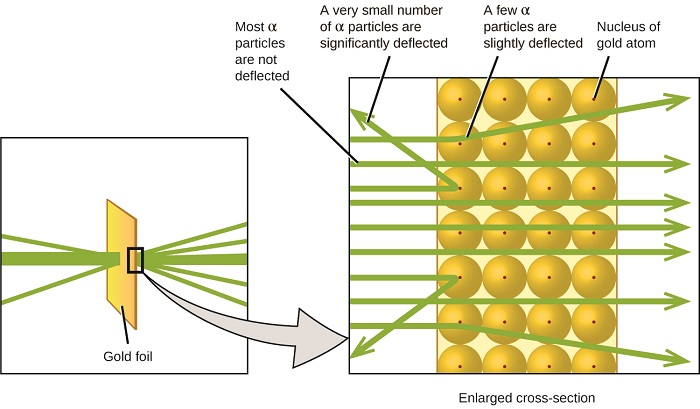
Discovery of the Nucleus
- Conclusion: Atoms have a tiny, dense, positive center
- The nucleus: Contains most of the atom's mass
- Empty space: Most of the atom is empty
- Electrons: Orbit far from the nucleus
Understanding Rutherford's experiment
- Alpha particles (positive) from radioactive source
- Very thin gold foil (only a few atoms thick)
- Fluorescent screen to detect where particles land
- Dark room to observe flashes of light
- ~99% of particles passed straight through → atoms mostly empty
- Some deflected at small angles → positive charges repelling
- ~1 in 8000 bounced back → hit something very dense and positive
- Size of nucleus: about 10,000 times smaller than the atom
- If the nucleus were a marble, the atom would be a football stadium
- Electrons would be like flies buzzing around the stadium
Niels Bohr's Model (1913)
Quantized Energy Levels
- Problem with Rutherford's model: Electrons should spiral into nucleus
- Bohr's solution: Electrons only exist in specific orbits
- Energy levels: Each orbit has a fixed energy
- Quantum jumps: Electrons jump between levels, absorbing or emitting light
Features of Bohr's Model
- Planetary model: Electrons orbit like planets around sun
- Discrete orbits: Only certain distances from nucleus allowed
- Energy emission: Light emitted when electrons drop to lower levels
- Hydrogen spectrum: Successfully explained hydrogen's spectral lines
Modern Quantum Mechanical Model (1920s-present)
Wave-Particle Duality
- De Broglie: Proposed electrons behave like waves
- Schrödinger: Developed wave equation for electrons
- Heisenberg: Uncertainty principle – can't know exact position and momentum
- Orbitals: Probability clouds where electrons likely exist
Key Concepts
- No fixed orbits: Electrons don't follow specific paths
- Probability distributions: We can only say where electrons probably are
- Electron clouds: Regions of high electron probability
- Complex shapes: s, p, d, f orbitals have different geometries
How Do We "See" Atoms?
Since atoms are incredibly tiny (about 10⁻¹⁰ meters, or one ten-billionth of a meter), we cannot see them with ordinary microscopes. Scientists have developed amazing technologies to visualize these tiny building blocks.
Why Can't We Use Ordinary Microscopes?
The Wavelength Problem
- Resolution limit: Can't see objects smaller than the wavelength used
- Visible light wavelength: 400-700 nanometers
- Atom size: About 0.1-0.3 nanometers
- Conclusion: Atoms are ~1000 times smaller than light wavelength!
Electron Microscopy
Transmission Electron Microscope (TEM)
- Developed by: Ernst Ruska (1930s, Nobel Prize 1986)
- Principle: Uses electron beams instead of light
- Electron wavelength: Much shorter than visible light
- Resolution: Can see individual atoms in some materials
Scanning Electron Microscope (SEM)
- Principle: Scans surface with focused electron beam
- Creates: 3D-like images of surfaces
- Used for: Surface structure analysis
- Resolution: Very high, but not quite atomic level
Scanning Probe Microscopy
Scanning Tunneling Microscope (STM)
- Invented by: Gerd Binnig and Heinrich Rohrer (1981, Nobel Prize 1986)
- Principle: Uses quantum tunneling effect
- How it works: A tiny needle scans very close to surface
- Resolution: Can image individual atoms!
- Famous image: IBM spelled with 35 xenon atoms (1989)
Atomic Force Microscope (AFM)
- Principle: Tiny tip "feels" the surface
- Measures: Forces between tip and atoms on surface
- Advantages: Works on non-conducting materials
- Applications: Biology, materials science, nanotechnology
How the Scanning Tunneling Microscope works
- Electrons can "tunnel" through barriers they shouldn't classically pass
- A sharp metal tip is brought extremely close to a surface (~1 nm)
- Electrons tunnel between tip and surface
- Tunneling current is extremely sensitive to distance
- Tip scans across the surface in a raster pattern
- Computer adjusts tip height to keep current constant
- Height adjustments map out the surface topography
- Individual atoms appear as bumps in the image!
- First time humans could "see" individual atoms
- Can also be used to move individual atoms
- Enabled nanotechnology and molecular manipulation
X-Ray Crystallography
How It Works
- Principle: X-rays diffract through crystal lattices
- Pattern analysis: Diffraction pattern reveals atomic arrangement
- Resolution: Can determine atomic positions precisely
- Famous use: Discovering DNA structure (Watson, Crick, Franklin)
The Scale of Atoms
Incredible Smallness
- Atom diameter: ~10⁻¹⁰ m (0.1 nanometer)
- Nucleus diameter: ~10⁻¹⁵ m (100,000 times smaller than atom!)
- If atom were a football stadium: Nucleus would be a pea at center
Mind-Boggling Numbers
- Atoms in a grain of sand: ~50,000,000,000,000,000,000 (5 × 10¹⁹)
- Atoms in human body: ~7 × 10²⁷
- Atoms in a drop of water: ~5 × 10²¹
Mostly Empty Space
- Nucleus contains: 99.9% of atom's mass
- Nucleus occupies: Only 1/10,000,000,000,000 of atom's volume
- If you removed all empty space from atoms: Entire human race would fit in a sugar cube!
Key Takeaways
- Atomic theory evolved from Greek philosophy to modern quantum mechanics
- Democritus first proposed atoms, but Dalton made it scientific
- Thomson discovered electrons and proposed the plum pudding model
- Rutherford discovered the nucleus through his gold foil experiment
- Bohr introduced quantized energy levels for electrons
- Atoms consist of protons, neutrons (in nucleus), and electrons (in cloud)
- Protons determine the element; neutrons determine the isotope
- We can "see" atoms using electron microscopes and scanning probe microscopes
- The STM can image and even manipulate individual atoms
- Atoms are incredibly tiny – mostly empty space with a dense nucleus