Loading history...
| Property | Metals 🧲 | Metalloids ⚗️ | Nonmetals 🌬️ |
|---|---|---|---|
| Electrical Conductivity | High | Medium (semiconductors) | Low |
| Thermal Conductivity | High | Medium | Low |
| Appearance | Shiny | Shiny or dull | Dull |
| Malleability | Yes | No | No |
| Ductility | Yes | No | No |
| State at Room Temperature | Mostly solid | Solid | Solid/liquid/gas |
| Electron Behavior | Lose electrons | Can gain or lose | Gain electrons |
| Examples | Fe, Cu, Al | Si, Ge | O, N, Cl |
Hydrogen
Hydrogen is truly unique - the simplest element with just one proton, yet it powers the stars and holds the promise of clean energy for our planet. As the most abundant element in the universe, hydrogen connects the smallest scale of atoms to the grandest scale of galaxies.
What Makes Hydrogen Special?
The Simplest Element
Basic Structure
- Atomic number: 1 (one proton)
- Electron configuration: 1s¹ (one electron)
- Atomic mass: 1.008 u (lightest element)
- Symbol: H (from Greek "hydro" = water, "genes" = maker)
Unique Position
- Periodic table placement: Often shown separately or in Group 1
- Neither metal nor non-metal: Unique properties
- Can lose or gain electrons: Forms H⁺ and H⁻ ions
- Forms diatomic molecules: H₂ in nature
Why hydrogen behaves so uniquely
Universal Presence
- Universe composition: ~75% by mass, 90% by number of atoms
- Stars: Main fuel for nuclear fusion
- Interstellar space: Most abundant element
- Big Bang: First element formed after the universe began
On Earth
- Earth's crust: 0.14% by weight
- Combined form: Mainly in water and organic compounds
- Free hydrogen: Very rare in atmosphere (<1 ppm)
- Biological importance: Essential component of all organic molecules
Physical Properties
Gaseous State
Standard Conditions
- State: Colorless, odorless gas
- Density: 0.089 g/L (lightest gas)
- Molecular form: H₂ (diatomic)
- Bond strength: Very strong H-H bond (436 kJ/mol)
Extreme Conditions
- Boiling point: -253°C (-423°F)
- Melting point: -259°C (-434°F)
- Critical point: -240°C, 13 atm
- Liquid hydrogen: Used as rocket fuel
Chemical Properties
Reactivity
- Generally unreactive: Strong H-H bond
- Combustible: Burns in air with almost invisible flame
- Reducing agent: Can remove oxygen from compounds
- Forms compounds: With most elements
Bonding Behavior
- Covalent bonding: Shares electrons with non-metals
- Ionic character: Can form H⁺ and H⁻ ions
- Hydrogen bonding: Weak attraction between molecules
- Metallic behavior: Under extreme pressure
Electronic structure effects:
Single electron:- No inner electrons to shield nuclear charge
- Very small size when forming H⁺
- Can approach other nuclei very closely
- Forms very strong bonds relative to its size
- Alkali metals: Have inner electrons, much larger
- Halogens: Seven electrons, different chemistry
- Hydrogen: Unique - can act like both
- H bonded to N, O, or F becomes partially positive
- Attracts lone pairs on other N, O, F atoms
- Much stronger than van der Waals forces
- Crucial for water, DNA, protein structure
Metals
Metals are elements that readily lose electrons to form positive ions and are characterized by their unique physical and chemical properties.
Position in Periodic Table
- Location: Left side and center of periodic table
- Main groups: Groups 1, 2, and 13-15 (partially)
- Transition metals: Groups 3-12
- Inner transition: Lanthanides and actinides
Key Physical Properties of Metals
- Luster: Shiny, reflective surfaces due to mobile electrons.
- Electrical conductivity: Excellent; used in wiring (silver, copper, aluminum).
- Thermal conductivity: Good heat transfer; used in cookware, radiators.
- Malleability: Can be hammered into sheets (e.g., gold leaf, aluminum foil).
- Ductility: Can be drawn into wires (e.g., copper, gold).
- Hardness and strength: Varies widely; the trend in each period is that they start soft and become harder in the middle of transition metals to be later softer again.

Why metals have these unique properties
- Metal atoms lose valence electrons
- Positive ions arranged in regular pattern
- Electrons form "sea" around positive ions
- Electrons are delocalized and mobile
- Conductivity: Mobile electrons carry current and heat
- Luster: Electrons interact with light and reflect it
- Malleability/Ductility: Non-directional bonding allows layers to slide
- Strength: Electrostatic attraction holds structure together
- Ionic compounds: Localized electrons, brittle
- Covalent compounds: Directional bonds, often brittle
- Metals: Unique combination of strength and flexibility
An alloy is a mixture of two or more elements, usually metals, made to improve properties like strength or resistance to corrosion. Example:
- Bronze: Copper + tin (harder and more durable than copper alone)
- Steel: Iron + carbon (stronger than pure iron)
- Brass: Copper + zinc (used in instruments and fittings)
Metalloids (Semimetals)
Metalloids, also called semimetals, are the remarkable elements that bridge the gap between metals and non-metals. These unique elements power our digital age, from the silicon in computer chips to the boron in advanced materials.
What Are Metalloids?
Definition and Characteristics
Physical Properties
Appearance
- Luster: Metallic or semi-metallic shine
- Color: Various - silicon (gray), boron (brown), arsenic (gray)
- Brittleness: Solid metalloids are brittle like non-metals
- Crystal structure: Well-defined crystalline forms
Electrical Properties
- Semiconductor behavior: Key characteristic
- Conductivity: Between metals and non-metals
- Temperature dependence: Conductivity increases with temperature
- Purity sensitive: Small impurities greatly affect conductivity
The importance of semiconductors
Semiconductors are materials with conductivity between metals and insulators, crucial for electronics. Their conductivity can be precisely controlled by adding small amounts of other elements (doping).
N-type: Doped with elements like phosphorus (extra electrons, negative charge carriers). P-type: Doped with elements like boron (creates "holes," positive charge carriers). P-N junctions (where N-type and P-type meet) allow current to flow in one direction—key for diodes, transistors, and solar cells.Semiconductor Properties
What Are Semiconductors?
Non-metals
Non-metals are essential elements that make up the air we breathe, the water we drink, and the organic molecules in our bodies. Despite being less numerous than metals, non-metals play crucial roles in chemistry, biology, and technology.
There is a huge variety of non-metals, from the gases that fill our atmosphere to the solids that form the basis of life. They are defined by their tendency to gain electrons and form negative ions, as well as their high electronegativity and ability to form covalent bonds.
Position in Periodic Table
- Location: Upper right area of periodic table
- Groups: Primarily Groups 14-18
Physical Properties of Non-metals
Poor Electrical Conductivity
- Reason: Electrons are locked in covalent bonds
- Exception: Graphite (carbon) conducts electricity since they create delocalized π electrons
Poor Thermal Conductivity
Brittleness
- Solid non-metals: Break rather than bend
Appearance
- Appearance: Dull, not shiny
- Colors: Various - yellow sulfur, red bromine, purple iodine
States of Matter
- Variety: Can be gases, liquids, or solids at room temperature
- Small compounds: Often gases (O₂, N₂)
- Larger molecules: Often solids (sulfur, phosphorus, diamonds)
Allotropes
Allotropes are different structural forms of the same element, with distinct physical and chemical properties. For example, carbon has several allotropes, including diamond (hard, insulating), graphite (soft, conductive), fullerenes (spherical molecules), and graphene (single layer of carbon atoms).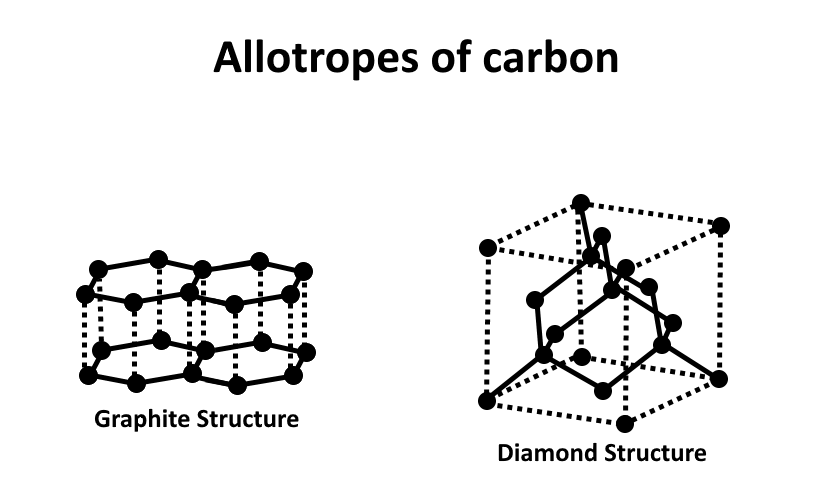
Noble Gases
They are the unreactive, noble elements of Group 18, with complete outer electron shells that make them stable and inert. Despite their lack of reactivity, noble gases have important applications in lighting, welding, and even breathing mixtures for deep-sea diving.
Usage:
- Protect things from reacting (like the case of argon in light bulbs)
- Lighting: Neon signs, argon in incandescent bulbs, xenon in high-intensity lamps
- Cooling: Liquid helium for superconducting magnets, liquid nitrogen for cryogenics