Loading history...
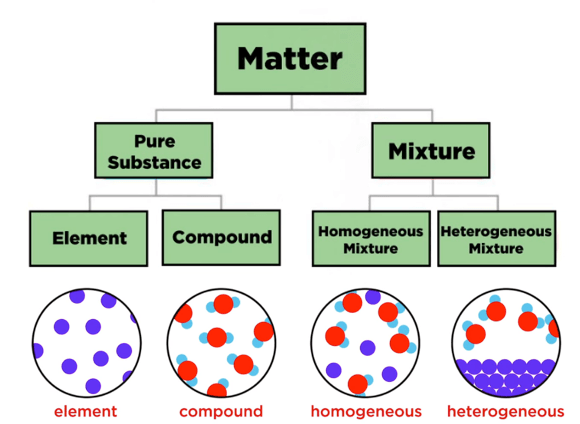
Everything around us is made of matter, and all matter is composed of elements and compounds. Understanding these fundamental building blocks helps us comprehend how the world works at the atomic level.
What Are Elements?
Definition and Characteristics
An element is a pure substance that consists of only one type of atom and cannot be broken down into simpler substances by chemical means.
Key Properties of Elements
- Pure substances: Made of only one type of atom
- Cannot be decomposed: Cannot be broken down chemically
- Unique identity: Each element has a unique atomic number
- Characteristic properties: Each has distinct physical and chemical properties
- Building blocks: Combine to form all other substances
What Are Compounds?
Definition and Formation
A compound is a pure substance formed when two or more different elements are chemically bonded together in fixed proportions.
Compounds are ussually referred as molecules, which are the smallest units of a compound that retain its properties. For example, a water molecule (H₂O) consists of two hydrogen atoms and one oxygen atom bonded together.
Key Properties of Compounds
- Multiple elements: Always contain two or more different elements
- Chemical bonds: Elements held together by chemical forces
- Fixed composition: Always same ratio of elements
- New properties: Properties completely different from constituent elements
- Can be decomposed: Broken down into elements by chemical methods
Chemical Formulas
Compounds are represented by chemical formulas that show:
- Element symbols: Which elements are present
- Subscripts: How many atoms of each element
- Proportions: Fixed ratios between elements
Reading Chemical Formulas
- H₂O: 2 hydrogen atoms + 1 oxygen atom = water
- CO₂: 1 carbon atom + 2 oxygen atoms = carbon dioxide
- NaCl: 1 sodium atom + 1 chlorine atom = table salt
- C₆H₁₂O₆: 6 carbon + 12 hydrogen + 6 oxygen = glucose
Comparing Elements and Compounds
| Property | Elements | Compounds |
|---|---|---|
| Composition | One type of atom only | Two or more different elements |
| Separation | Cannot be broken down chemically | Can be decomposed into elements |
| Properties | Characteristic of that element | Different from constituent elements |
| Examples | H, O, C, Fe, Au | H₂O, CO₂, NaCl, C₆H₁₂O₆ |
| Formation | Cannot be created from simpler substances | Formed by chemical combination of elements |
| Symbols/Formulas | Single element symbol (e.g., O) | Chemical formula (e.g., H₂O) |