Loading history...
Chemical Thermodynamics
Chemical thermodynamics is the study of energy changes that accompany chemical reactions. During a chemical reaction, chemical bonds of the reactants are broken and new bonds are formed to create products. These processes involve the absorption and release of energy.Energy in Reactions
Every chemical substance has a certain amount of internal chemical energy. When a chemical reaction occurs, the total energy of the products is usually different from the total energy of the reactants. This difference is exchanged with the surroundings, usually in the form of heat.
Exothermic Reactions
An exothermic reaction releases energy to the surroundings.
- Energy is released (often as heat, light, or sound).
- The surroundings become warmer.
- The products have less chemical energy than the reactants.
- The change in energy (enthalpy, ΔH) is negative (ΔH < 0).
- Combustion: Burning of fuel (like wood or gas) releases heat and light.
- Respiration: Cells in our body break down glucose to release energy.
- Neutralization: Mixing an acid and a base usually releases heat.
Example: Combustion reactions
Endothermic Reactions
An endothermic reaction absorbs energy from the surroundings.
- Energy is absorbed from the surroundings.
- The surroundings become cooler.
- The products have more chemical energy than the reactants.
- The change in energy (enthalpy, ΔH) is positive (ΔH > 0).
- Photosynthesis: Plants absorb sunlight energy to convert water and carbon dioxide into glucose.
- Thermal decomposition: Heating limestone (calcium carbonate) to break it down.
- Cold packs: Dissolving ammonium nitrate in water absorbs heat, making the pack feel very cold.
Example: In an instant cold pack
> We don't normally consider the dissolution of salts in water to be a chemical reaction, but it is a good example of an endothermic process.
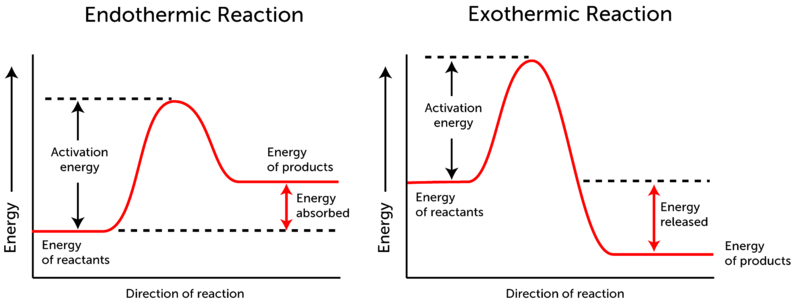
Energy Diagrams
An energy diagram (or potential energy diagram) is a graph that shows the progress of a chemical reaction and the changes in energy that occur.
- y-axis: Represents the potential energy of the substances.
- x-axis: Represents the progress of the reaction (from reactants to products).
- Activation Energy (\(E_a\)): The minimum energy "hill" that must be overcome for the reaction to start. Even exothermic reactions need a little energy push to get started (like a spark for a fire).
Energy Diagram for an Exothermic Reaction
In an exothermic reaction, the reactants start with more energy than the products end up with. The difference in energy is released.
- The curve starts high (energy of reactants).
- It goes up to a peak (activation energy).
- It drops down to a level lower than the starting point (energy of products).
- The overall energy change (ΔH) is negative.
Energy Diagram for an Endothermic Reaction
In an endothermic reaction, the reactants start with less energy than the products end up with. Energy must be continuously supplied to keep the reaction going.
- The curve starts low (energy of reactants).
- It goes up to a peak (activation energy).
- It drops down to a level higher than the starting point (energy of products).
- The overall energy change (ΔH) is positive.