Loading history...
Stoichiometry
Stoichiometry is the study of the quantitative relationships between reactants and products in chemical reactions.Balanced Chemical Equations
A balanced equation shows the correct ratio of reactants to products, following the Law of Conservation of Mass (atoms are neither created nor destroyed).
Example:- 2 molecules of hydrogen react with 1 molecule of oxygen
- to produce 2 molecules of water
- The mole ratio is 2 : 1 : 2
How to Balance Equations
There are different methods to balance chemical equations:
1. Inspection Method (Trial and Error)
- Count atoms of each element on each side of the equation.
- Adjust coefficients (never change the subscripts!) to balance the atoms.
- Start with the most complex molecule.
- Leave pure elements (like \(O_2\) or \(H_2\)) for last.
Example: Balance the equation: C + O₂ → CO₂
Left side: 1 C, 2 O.
Right side: 1 C, 2 O.
Answer: C + O₂ → CO₂ (Already balanced!)
Example: Balance: Fe + O₂ → Fe₂O₃
We start with O. Left has 2, right has 3. The common multiple is 6.
Place a 3 in front of O₂ and a 2 in front of Fe₂O₃.
Now we have 4 Fe on the right, so put a 4 in front of Fe on the left.
Answer: 4Fe + 3O₂ → 2Fe₂O₃
2. The Oxygen Fraction Trick (for Combustion)
When balancing combustion reactions with \(O_2\), you can use a fraction \(n/2\) to get an odd number of oxygen atoms, and then multiply the entire equation by 2 to get whole numbers.
Example: Balance the combustion of Butane: C₄H₁₀ + O₂ → CO₂ + H₂O
Balance C first: C₄H₁₀ + O₂ → 4CO₂ + H₂O
Balance H next: C₄H₁₀ + O₂ → 4CO₂ + 5H₂O
Count Oxygen on the right: (4 × 2) + 5 = 13 O atoms.
To get 13 O atoms on the left, use the fraction 13/2 for O₂:
C₄H₁₀ + 13/2 O₂ → 4CO₂ + 5H₂O
Multiply everything by 2 to remove the fraction:
Answer: 2C₄H₁₀ + 13O₂ → 8CO₂ + 10H₂O
3. Algebraic Method
For complex equations, use variables for coefficients and solve the system of linear equations.
- Assign a letter (a, b, c, etc.) to each coefficient.
- Write an algebraic equation for each element based on the conservation of atoms.
- Assign a value of 1 (or 2) to the variable that appears most often.
- Solve for the other variables.
- If there are fractions, multiply all variables by the lowest common denominator to get integers.
Example: Balance: a NH₃ + b O₂ → c NO + d H₂O
Equations for each element:
- N: a = c
- H: 3a = 2d
- O: 2b = c + d
Let's set a = 2 (to make 3a an even number, avoiding fractions for d).
If a = 2, then c = 2.
From H: 3(2) = 2d → 6 = 2d → d = 3.
From O: 2b = c + d → 2b = 2 + 3 → 2b = 5 → b = 5/2.
Our coefficients are: a=2, b=5/2, c=2, d=3.
Multiply all by 2 to remove the fraction: a=4, b=5, c=4, d=6.
Answer: 4NH₃ + 5O₂ → 4NO + 6H₂O
Stoichiometric Calculations
Use the balanced equation to convert between quantities of reactants and products. The coefficients tell us the exact mole ratio.
General steps:- Write the balanced equation.
- Convert the given amount into moles (if it isn't already).
- Use the mole ratio to find the moles of the desired substance.
- Convert those moles into the desired units (grams, particles).
Example: Given the reaction: 2H₂ + O₂ → 2H₂O. How many grams of water are produced from 4 mol of hydrogen?
We are given 4 mol of H₂. We want mass of H₂O.
The mole ratio from the equation is 2 mol H₂ : 2 mol H₂O (which is 1:1).
Calculate moles of H₂O: 4 mol H₂ × (2 mol H₂O / 2 mol H₂) = 4 mol H₂O.
Convert moles of H₂O to mass: 4 mol × 18 g/mol = 72 g.
Answer: 72 g
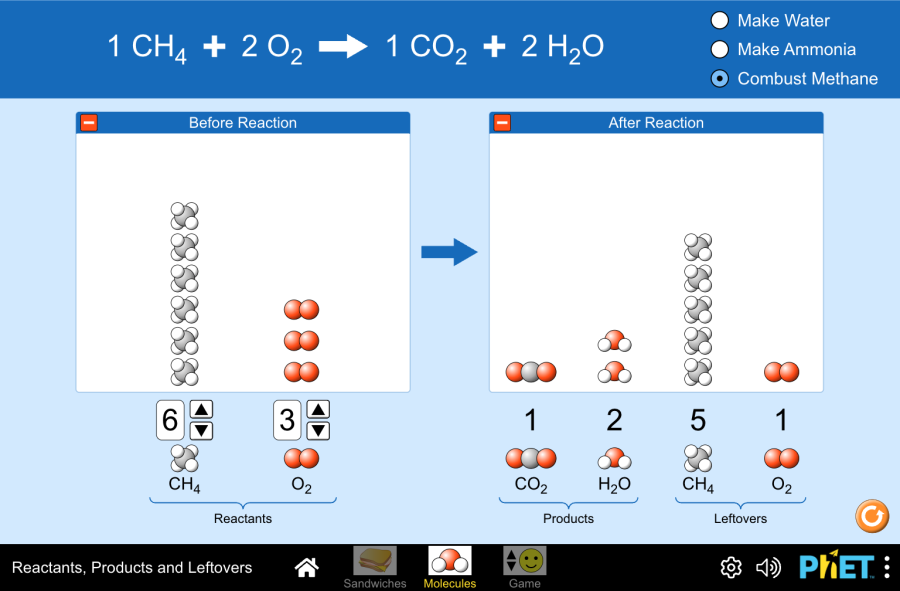
Example: Calculate the mass of CO₂ produced when burning 16 g of CH₄ in excess oxygen: CH₄ + 2O₂ → CO₂ + 2H₂O
Molar mass CH₄ = 12 + 4(1) = 16 g/mol.
Molar mass CO₂ = 12 + 2(16) = 44 g/mol.
Convert mass of CH₄ to moles: 16 g ÷ 16 g/mol = 1 mol CH₄.
Use mole ratio: 1 mol CH₄ produces 1 mol CO₂.
Convert moles of CO₂ to mass: 1 mol CO₂ × 44 g/mol = 44 g.
Answer: 44 g
Example: How many moles of Aluminum are needed to react with 3 moles of HCl? 2Al + 6HCl → 2AlCl₃ + 3H₂
We are given 3 mol of HCl. We want moles of Al.
The mole ratio is 2 mol Al : 6 mol HCl.
Calculate: 3 mol HCl × (2 mol Al / 6 mol HCl) = 1 mol Al.
Answer: 1 mol Al
Limiting Reactant
Often, reactants are not mixed in exact stoichiometric ratios. Since all reactants are needed for a reaction to occur, the one that runs out first—the limiting reactant—determines how much product can be formed. It is completely consumed while the others remain in excess.